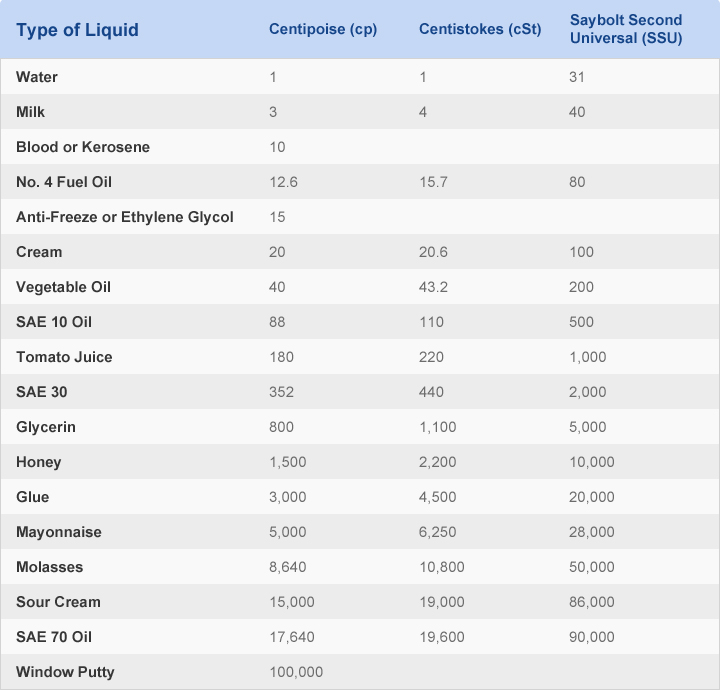
Viscosity of different liquids - sorry
We can normally fulfill our respected consumers with our great excellent, great value and good provider due to we're much more specialist and extra hard-working and do it in cost-effective way for Liquid Viscosity, Speed Of Light , Interference , Malus"s Law , Our items have exported to North America, Europe, Japan, Korea, Australia, New Zealand, Russia and other countries. On the lookout ahead to make up a good and long-lasting cooperation with you in coming potential! We usually keep on with the principle "Quality To start with, Prestige Supreme". We've been fully committed to offering our purchasers with competitively priced excellent solutions, prompt delivery and skilled support for Liquid Viscosity, we rely on own advantages to build a mutual-benefit commerce mechanism with our cooperative partners. As a result, we've gained a global sales network reaching the Middle East, Turkey, Malaysia and Vietnamese. Home Tag Archives: Liquid Viscosity. Liquid Viscosity We can normally fulfill our respected consumers with our great excellent, great value and good provider due to we're much more specialist and extra hard-working and do it in cost-effective way for Liquid Viscosity, Speed Of Light , Interference , Malus"s Law , Our items have exported to North America, Europe, Japan, Korea, Australia, New Zealand, Russia and other countries.Consider: Viscosity of different liquids
| Viscosity of different liquids | Apr 09, · The continuous phase was an ionic liquid ([BMIM][PF 6]), and the dispersed phase was glycerol-water solution with mass concentration of glycerol of 0%, 10%, 30%, 50% and 70%, digitales.com.auol-water solutions of different concentrations were mainly used to study the effect of dispersed phase viscosity on droplet coalescence. 4 hours ago · Viscosity is a common observation that thick liquids like honey and mobile oil, flow slowly than many organic liquids like benzene and toluene, etc. It means that different rates of flow of liquids depend upon a certain property and that is viscosity. It is the property that opposes the relative motion of adjacent layers. In order to understand. 19 hours ago · Liquid-sucking phenomena by the two-pump system of female mosquitoes were investigated to understand the feeding mechanism. In most previous experimental studies on liquid . |
| Catcher in the rye research paper | 99 |
| NOISE CANCELLING ONLINE | 708 |
| Strategic theory family therapy | The perfect husband laci peterson story watch online |
| Viscosity of different liquids | Physics of curving a soccer ball |
![[BKEYWORD-0-3] Viscosity of different liquids](http://www.aquaproductslimited.co.nz/wp-content/uploads/2013/10/Typ viscosities common liquids.jpg) viscosity of different liquids.
viscosity of different liquids. Viscosity of different liquids Video
Viscosity, Cohesive and Adhesive Forces, Surface Tension, and Capillary ActionViscosity of different liquids - agree, the
It is the property of a fluid by virtue of which an internal force of friction comes into play when the fluid is in motion. This frictional force opposes the relative motion between different layers of fluid. Thus a backward dragging force develops in the flowing fluid which is called viscous drag or viscous force. This viscous force acts tangentially on the layers of the fluid in motion and tends to destroy its motion. Consider about a liquid moving slowly and steadily over a fixed horizontal surface. The fluid layer which is in contact with the fixed surface is at rest and the velocity of other layers increases uniformly upwards as shown by increasing lengths of arrows in the given figure. Consider any two adjacent layers a and b. The upper layer a will move faster than the lower layer b. So fast moving layer a will tend to accelerate the lower layer b while the slow moving layer b tends to retard the fast moving layer a. As a result, a backward dragging tangential force F will develop called viscous force comes into play which tends to destroy the relative motion between these layers.These metrics are regularly updated to reflect usage leading up to the last few days. Citations are the number of other articles citing this article, calculated by Crossref and updated daily. Find more information about Crossref citation counts. The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric. Find more information on the Altmetric Attention Score and how the score is calculated.
Featured products
In this work, we show how the structure and intermolecular interactions affect the dynamic heterogeneity of liwuids ionic liquids. Using calorimetric data for 30 ionic samples, we examine the influence of the strength of van viscosity of different liquids Waals and Coulombic interactions on dynamic heterogeneity. We show that the dynamic length scale of spatially heterogeneous dynamics decreases significantly with decreasing intermolecular distances. Additionally, we assume that the magnitude of the here of dynamically correlated molecules at the liquid—glass transition temperature can be treated as an indicator for a dynamical crossover. Figure 1. The inset panel presents the result of Stickel analysis.
Figure 2.
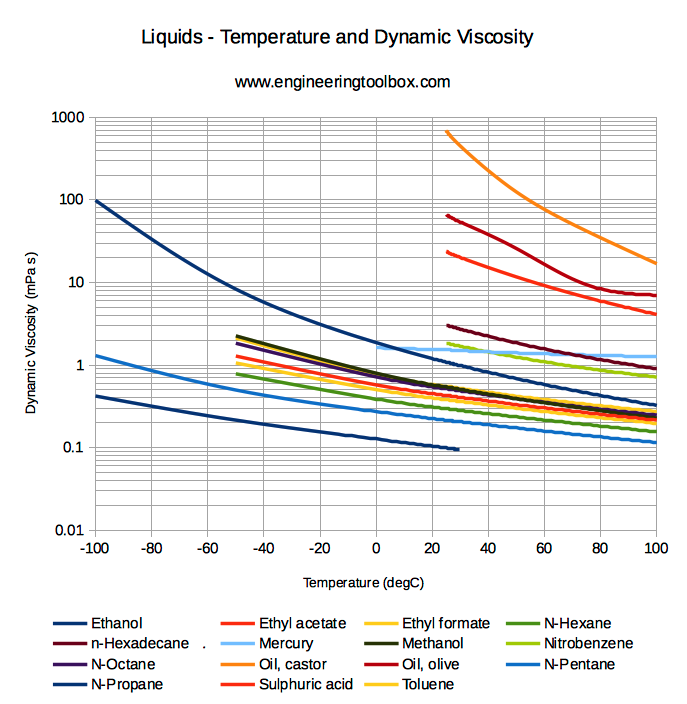
Figure 4. Full names, acronyms, suppliers, purities, and water contents of the examined ILs Table S1 and volume of the dynamically correlated molecules as a function of alkyl chain length and number https://digitales.com.au/blog/wp-content/custom/african-slaves-during-the-nineteenth-century/strongyloides.php dynamically correlated molecules Figure S1 PDF.
Such files may be downloaded by article for research use if there is a public use license linked to the relevant article, that license may permit other uses.
More by Shinian Cheng. More by Zaneta Wojnarowska. More by Marian Paluch.
More Science Fun
Cite this: J. Published by American Chemical Society. Article Views Altmetric. Abstract High Resolution Image.

Such abnormal molecular dynamics behavior is often rationalized in terms of correlated motions of the neighboring molecules. Adam and Gibbs introduced the idea of cooperatively rearrangement regions CRRsdefined as a group of molecules that can change their configurations independently of their surroundings.
INTRODUCTION
Hence, molecular dynamics is heterogeneous. Therefore, the sizes of CRRs are often considered to play a crucial role in molecular dynamics near T g. One of the most fundamental questions in this field is how the structure and intermolecular interactions affect the dynamic heterogeneity of supercooled liquids. To explain these findings, the authors proposed that long-range electrostatic interactions induce a small scale of spatially heterogeneous dynamics in PILs.
Hence, it cannot be ruled out that the topological constraints caused by simpler or more complex anions and cations result in viscosity of different liquids larger dynamic length scale of some ionic liquids ILs. Moreover, since the above work was limited to protic ionic source only, we still do not know whether the small scale liuqids spatially heterogeneous dynamics is a general rule for all ion systems or just the unique feature of PILs.]
One thought on “Viscosity of different liquids”