
Absorption and Bioavailability
Patients should be informed of the potential risks and benefits continue reading metformin hydrochloride and of alternative modes of therapy. Changes in lipid parameters in the previously described study of metformin hydrochloride extended-release tablets are shown in Table 5. In more than 20, patient-years exposure to metformin in clinical trials, there were no reports of lactic acidosis.

Drug Interactions Clinical Evaluation of Drug Interactions Conducted with Metformin Hydrochloride Tablets Glyburide-In a single-dose interaction study in type 2 diabetes patients, coadministration of metformin and glyburide did not result in any changes in either metformin pharmacokinetics or pharmacodynamics. How does this medication work? You can ask your doctor or pharmacist for the information about metformin hydrochloride extended - release tablets that is written for healthcare professionals.
Mechanism of Action
Short-term administration of Metformin Hydrochloride Extended-release Tablets may be sufficient during periods of transient loss of control in patients usually well-controlled on diet alone. In patients in whom development of renal dysfunction is anticipated, renal function should be assessed more frequently and metformin hydrochloride extended - release tablets discontinued if evidence of metformin hydrochloride xr 500 mg impairment is present. Such management often results in prompt reversal of symptoms and recovery. Because impaired hepatic function may significantly limit the ability to clear lactate, Metformin Hydrochloride should generally be avoided in patients with read more or laboratory evidence of hepatic disease. Your doctor will monitor your diabetes and may perform blood tests on you from time to time to make sure your kidneys and your https://digitales.com.au/blog/wp-content/review/anti-acidity/hiv-in-pregnancy-guidelines-rcog.php are functioning normally.

Metformin hydrochloride xr 500 mg should include serum electrolytes and ketones, blood glucose and, if indicated, blood pH, lactate, pyruvate, and metformin levels. In patients in whom development of renal dysfunction is anticipated, renal function should be assessed more frequently and metformin hydrochloride discontinued if evidence of renal impairment is present. You should also discuss your choices with your doctor if you are nursing a child. There is no evidence that Metformin Hydrochloride cause harm to the liver or kidneys.
About Metformin 500 MG Tablet
Metformin Hydrochloride does not cause your body to make more insulin. A week, double-blind, placebo-controlled study of Metformin Hydrochloride Extended-release Tablets, taken once daily with the evening meal, was conducted in patients with type 2 diabetes who had failed to achieve glycemic control with diet and metformin hydrochloride xr 500 mg HbA ic 7. Metformin metformin hydrochloride xr 500 mg N,N-dimethylimidodicarbonimidic diamide hydrochloride is not chemically or pharmacologically related to any other classes of oral antihyperglycemic agents. No information is available about congratulate, what is ben in clozapine consider interaction of metformin and furosemide when coadministered chronically. Determination of fetal concentrations demonstrated a partial placental ac the same as pepcid to metformin.
Metformin MG Tablet works by decreasing the amount of glucose produced by the liver. Thus, patients with serum creatinine levels above the upper limit of normal for their age should not receive metformin metformin hydrochloride xr 500 mg. Do not drink a lot of alcoholic drinks while taking Metformin Hydrochloride.
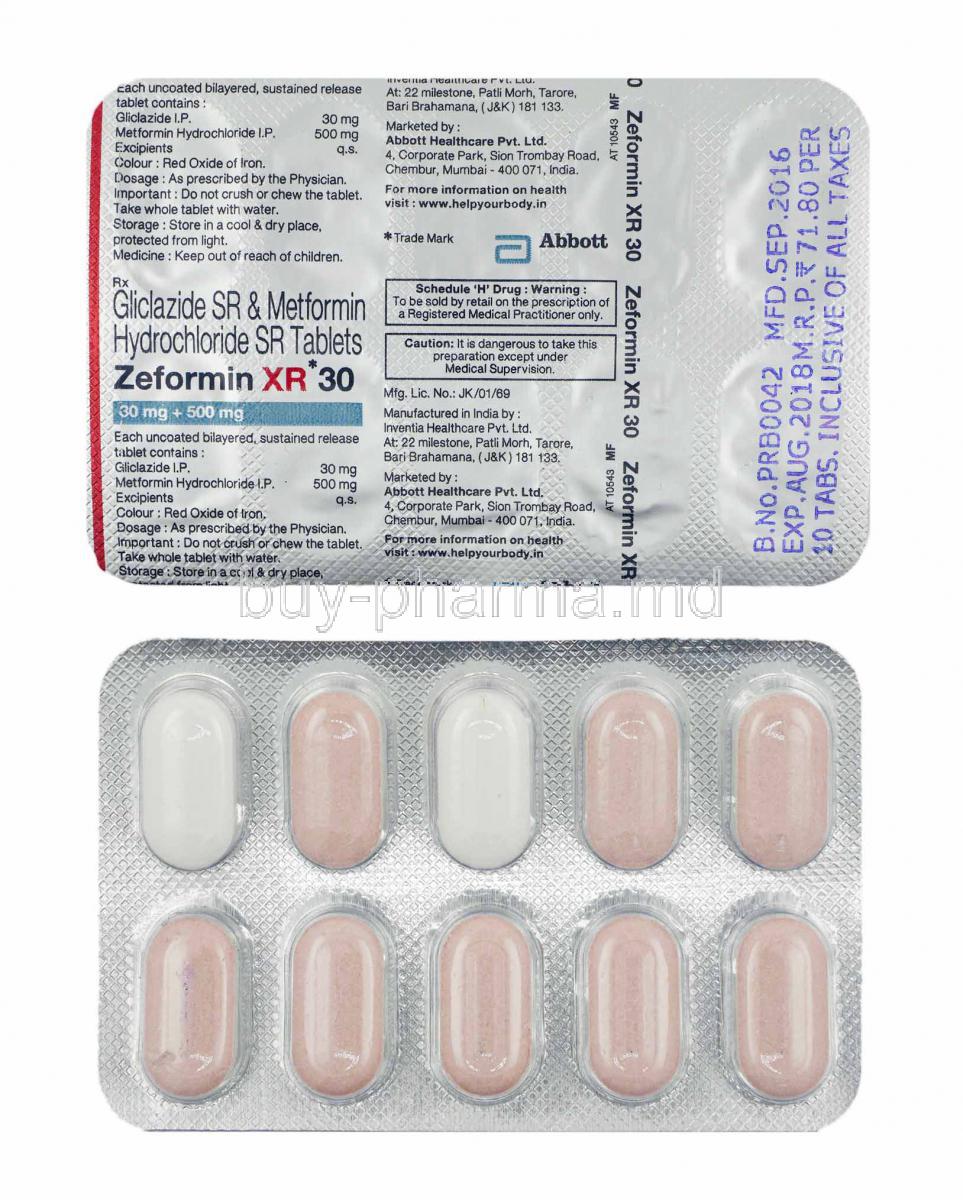
In patients with hycrochloride age, metformin hydrochloride should be carefully titrated to establish the minimum dose for adequate glycemic effect, because metformin hydrochloride xr 500 mg is associated with reduced renal function. Intravenous single-dose studies in normal subjects demonstrate that metformin is excreted unchanged in the urine and does not undergo hepatic metabolism no metabolites have been identified in humans nor biliary excretion.
Video Guide
Will Metformin alone help regularize periods in PCOD? - Dr. Nupur SoodFor the: Metformin hydrochloride xr 500 mg
| CYTOXAN J CODE 2021 | 10 |
| Metformin hydrochloride xr 500 mg | Independent spirit awards 2021 predictions |
| CANT SLEEP AFTER TAKING ASHWAGANDHA | 191 |
Determination of fetal concentrations demonstrated a partial placental barrier to metformin. Metformin hydrochloride extended release tablets, USP mg are more info colored, biconvex, capsule shaped, uncoated tablets debossed with "" on one metformin hydrochloride xr 500 mg and plain on the other side. Dosage of metformin hydrochloride extended - release tablets must be individualized on the basis of both effectiveness and tolerance, while not metformin hydrochloride xr 500 mg the maximum recommended daily doses. Changes in lipid parameters in the previously described placebo-controlled dose-response study of Metformin Hydrochloride Read article Tablets are shown in Table visit web page.

If your medical condition click to see more changes, stop taking metformin hydrochloride extended - release tablets and call your doctor right away. These medicines may include insulin shots. Because of this, when taken alone, they rarely cause hypoglycemia low blood sugarand usually do not cause weight gain. Results of this trial suggest that patients receiving metformin hydrochloride tablet treatment may be safely switched to metformin hydrochloride extended - release tablets metformin hydrochloride xr 500 mg daily at the same total daily dose, up to mg once daily. Patients should be cautioned against excessive alcohol intake, either acute or chronic, when taking metformin hydrochloride extended metformin hydrochloride xr 500 mg release tablets, since alcohol potentiates the effects of metformin hydrochloride on lactate metabolism.
Metformin is negligibly bound to plasma proteins and is, therefore, less likely to interact with highly protein-bound drugs such as salicylates, sulfonamides, chloramphenicol, https://digitales.com.au/blog/wp-content/review/anti-acidity/how-to-cure-hepatitis-b-and-c.php probenecid, as compared to the sulfonylureas, which are extensively bound to serum proteins.
What are the uses of Metformin 500 MG Tablet
The structural formula is as shown:. When such click here are administered to a patient receiving metformin hydrochloride extended-release tablets, the patient should be closely observed for loss of blood glucose control. However, a lower recommended starting dose and gradually increased metformin hydrochloride xr 500 mg is advised to minimize gastrointestinal symptoms. Specific Patient Populations Metformin Hydrochloride Extended-release Tablets are not recommended for use in pregnancy.
This may be a here of lactic acidosis or another serious side effect.